Table Of Content

To illustrate the effect of these progenitor cells on the rate of neurogenesis, we model and contrast the outcomes in neuronal number from developmental schemes that utilize OSVZ proliferation versus ones that do not. In every case, we start with two radial glial (RG) cells and plot neuron number over the course of eight cell cycles. The identification of the VZ as a site of asymmetric divisions and the SVZ as a site of symmetric divisions altered hypotheses regarding neocortical expansion. Independently of RG divisions, the number of symmetric IP cell divisions could also affect neuron number and be a determinant of neocortical size. Furthermore, expansion of founder cells within the VZ can only account in part for the neuron number observed in gyrencephalic species, as neocortical surface area is disproportionately expanded compared to the ventricular surface area, which remains relatively modest.
'Princess Leia' brainwaves help sleeping brain store memories - PsyPost
'Princess Leia' brainwaves help sleeping brain store memories.
Posted: Tue, 15 Nov 2016 08:00:00 GMT [source]
Increased Neurogenesis and Progenitor Cell Diversity

These observations suggest that oRG cells, although infrequent in some species, are part of a conserved developmental mechanism that can be studied in mice. Studies of the human brain and comparisons of developmental proliferative zones between species may ultimately help to explain what makes the human brain unique. It is commonly thought that the exceptional cognitive abilities of humans are related to the large size of the neocortex. Recent evidence has shown that primates have greater neuronal density (neuron number/brain mass) compared to rodents of equal brain mass, a feature that is likely related to the topological differences in foldedness that could have been influenced by OSVZ proliferation.
How unique is the human neocortex?
A reduction in functional cPCDH expression causes a lateral clustering of clonally related excitatory neurons originating from the same neural progenitor and a significant increase in synaptic connectivity. By contrast, overexpression of a single cPCDH isoform leads to a lateral dispersion of clonally related excitatory neurons and a considerable decrease in synaptic connectivity. These results suggest that patterned cPCDH expression biases fine spatial and functional organization of individual neocortical excitatory neurons in the mammalian brain. The studies summarized here suggest that an increase in sub-ventricular neurogenic divisions underlies the dramatic increase in neuron number in the evolutionary path to the human cerebral neocortex. In particular, oRG cells are the self-renewing building blocks of OSVZ proliferation and function to enhance both traditional roles of RG.
Analysis of Gene Expression in Human Brain
Β-catenin (CTNNB1) has also been established as a central regulator of proliferation versus differentiation in rodent neural progenitor cells. Β-catenin protein lies functionally at a crossroad between tissue architecture and regulation of the transcriptional activity that modifies cell fate, having dual roles as a component in the adherens junctions that link neuroepithelial/RG cells and as a transcriptional coactivator in the presence of Wnt signal. A later study tested the cell-autonomous role of β-catenin transcriptional activation by expressing ICAT or a dominant-negative version of TCF4, either of which blocks transcriptional activity by interfering with β-catenin/TCF4 complex formation (Woodhead et al., 2006). In this context, whereby neighboring cells provided a relatively normal radial scaffold, RG differentiated into neurons and migrated to the cortical plate, implying that β-catenin signaling suppresses neuronal differentiation and maintains progenitor cell proliferation independently of its role in adhesion. Observed increases in neuronal differentiation caused by β-catenin loss of function were preceded by increases in IP cell number, suggesting that β-catenin signaling restrains the development of IP cells from RG (Mutch et al., 2010). The divisions of oRG cells coupled with mitotic somal translocation begin to explain the radial growth of the OSVZ, as we can infer that oRG cells originate from the VZ, pass through the ISVZ, and leave a trail of daughter progenitor cells as they translocate basally.
However, the fibers provided by oRG cells are presumably still an important part of the scaffold for migrating neurons, functioning to guide cells part of the way toward the cortical plate. Dynamic imaging of oRG cell divisions in cultured slices of fetal neocortex shows that oRG cells exhibit a distinctive behavior whereby the cell soma rapidly ascends along the radial fiber during the hour preceding cell division, a process termed mitotic somal translocation (Figure 2B) (Hansen et al., 2010). This is in contrast to vRG cells, which undergo interkinetic nuclear migration and descend to the ventricle to undergo mitosis.
Regulation by Notch Signaling
An important insight came when HES1 expression was found to oscillate at 2–3 hr intervals in cultured cells due to negative autoregulation of Hes1 transcription (Hirata et al., 2002). Shimojo et al., (2008) discovered that HES1 control in RG is multifaceted, with the autonomous HES1 oscillatory circuit layered into a more pronounced level of regulation. HES1 protein was detected only during the phases of the cell cycle (late G1 through G2) when the cells were in the basal part of the VZ, but not during mitosis through early G1, when the cells were immediately adjacent to the ventricle. Furthermore, the oscillatory expression of HES1 in RG controls NEUROG2 and DLL1 expression and leads to their oscillation with inverse correlation. Therefore, RG are poised to generate neuronal progeny during periods of low HES1 expression.
Evolution
These are areas found in all studied primates.56 Likewise, there has been an expansion of motor cortex. Most mammals do not pay these costs, as they have smaller brains with less, or much less, neocortex, with less computational ability, which is balanced by earlier and often higher rates of reproduction, and typically reduced requirements for high grade food. From the examination of multiple species, a rough correlation can be made between the generation of three-layered, six-layered, and gyrencephalic neocortex and specific features of progenitor cell organization during development. The cortex of reptiles consists of three layers (Ulinski, 1974; Goffinet, 1983; Cheung et al., 2007) comparable to layers I, V, and VI in mammals, which are generated by a reptile homolog of RG, an ependymoradial glial cell (Weissman et al., 2003). Ependymoradial glia form a ventricular neuroepithelium, and there is no SVZ in the developing reptile dorsal cortex (Cheung et al., 2007).
Public Health Service and private philanthropic organizations that provided funding over the past four decades including NINDS, NEI, NIMH, NIDA, March of Dimes, NARSAD, NAAR and MOD Foundations and the Kavli Institute for Neuroscience at Yale. This collaborative Perspective, the result of a workshop held in 2023, proposes a set of community actions to increase the visibility of the developmental biology field. The authors make recommendations for new funding streams, frameworks for collaborations and mechanisms by which members of the community can promote themselves and their research.
“Princess Leia” Brainwaves Help Sleeping Brain Store Memories - Neuroscience News
“Princess Leia” Brainwaves Help Sleeping Brain Store Memories.
Posted: Tue, 15 Nov 2016 08:00:00 GMT [source]
Furthermore, developmental similarities between the human and ferret show that increased OSVZ proliferation and oRG cells are not primate-specific features (Figure 7A). The percentage of progenitor cells in the SVZ/OSVZ of rodents, carnivores, ungulates, and primates shows a remarkable positive correlation with the degree of neocortical gyrification (Reillo et al., 2010). Thus, development of OSVZ proliferation appears to be an important general feature for increasing neuronal number and neocortical surface area throughout Eutheria.
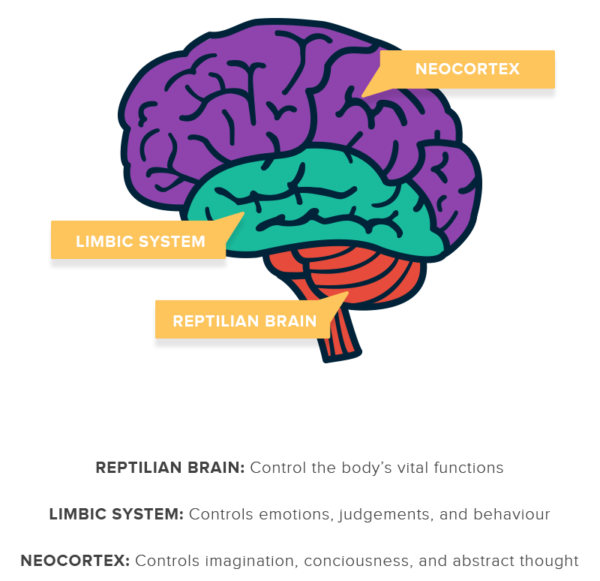
The neocortex is the largest part of the mammalian brain and is the seat of our higher cognitive functions. This outstanding neural structure increased massively in size and complexity during evolution in a process recapitulated today during the development of extant mammals. Accordingly, defects in neocortical development commonly result in severe intellectual and social deficits.
So you have this big sheet of cells that’s responsible for everything, it looks the same, pretty much the same everywhere. If we want to think about intelligence, our ability to understand the world and see the world, there’s some parts that are more important than others. And I have to be very, I’m going to annoy some neuroscientists in this answer, because everyone cares about their little piece, but ’m gonna give the sort of big picture. Okay, well, so you know, the human nervous system is a complex thing.
Vernon Mountcastle went further and he said the neocortex is divided in what are called columns and mini-columns. And I worked on the first mobile computers and the first smartphones. But I really had to get myself out of it, because I wanted to get back to brains. I think you could say what we’re doing is reverse engineering the brain. But you know, what it means is we have this thing in front of us, we know it works, we have a lot of data on it.
Dyes applied to the pial surface can diffuse into a proportion of oRG cells in the OSVZ through their basal fibers (Hansen et al., 2010). However, oRG cell fibers labeled from the OSVZ may not all reach the pia because cells born locally are unlikely to grow basal fibers that traverse such a great distance. The same may be true of vRG cells that initially gave rise to oRG cells but did not inherit the basal fiber. Dye and genetic labeling studies in the ferret have recently shown that radial fibers originating in the VZ diverge and form a fanned array by the time that they reach the pial surface.
By examining the evolutionary history of these loci, coding and regulatory mutations in genes likely to have strong effects on human cortical development can be identified. Using this approach, Walsh’s group identified a regulatory region necessary for proper folding of the perisylvian gyrus in humans and demonstrated that the activity of this regulatory region has changed across mammals (Fig. 2B). The gene Foxp2 was also first identified by a similar genetic mapping approach as being necessary for normal language development, and Svante Pääbo provided an update on the functional significance of two human-specific amino acid substitutions in the gene (Fig. 2D).
The ideas that oRG cells expand in number within the OSVZ and that OSVZ proliferation correlates with gyrus formation are substantiated by experimental evidence in the ferret (Reillo et al., 2010). Green fluorescent protein (GFP)-expressing retrovirus was delivered to the OSVZ of early postnatal ferret kits by stereotactic injection, followed by morphological analysis of labeled cells 2 days later. Because retrovirus labels only one of the two daughter cells following the initial cell division and because more than half of the labeled cells were reported to have oRG cell morphology, this ratio suggested a predominance of divisions in the OSVZ that result in two oRG cells (Reillo et al., 2010).
The expression patterns of Delta ligand (Campos et al., 2001) and Notch effector (Ohtsuka et al., 2006) suggested a scheme whereby Delta expressed on neuronally committed cells in the SVZ signals back to Notch receptors expressed on RG. This idea was tested by the removal of Mind-bomb1 (MIB1) (Yoon et al., 2008), an E3 ubiquitin ligase that is essential for generating functional Notch ligands (Itoh et al., 2003; Koo et al., 2005). MIB1 was expressed in neuronally committed TBR2-positive cells, and similar to studies of CBF1 deletion, its conditional knockdown in the neocortex also results in premature neuronal differentiation and depletion of RG cells. That isolated IP cells and postmitotic neurons stimulate HES1 activity in Notch-expressing neighboring cells in vitro further supports this idea. This study also raised the interesting possibility that the amount of time an IP cell pauses in the VZ before dividing or migrating away, a behavior that has been described as “sojourning” (Bayer and Altman, 1991; Noctor et al., 2004), could be related to its role in sustaining Notch activity in local RG cells. The neocortex is the most developed in its organisation and number of layers, of the cerebral tissues.[5] The neocortex consists of the grey matter, or neuronal cell bodies and unmyelinated fibers, surrounding the deeper white matter (myelinated axons) in the cerebrum.